 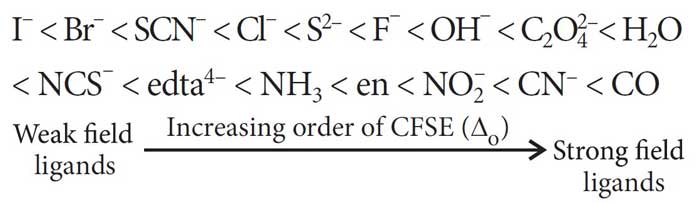
However, in this case, we will only consider crystal field theory and transition metal complexes.Ĭrystal field theory explains the bonding in complex ions in terms of electrostatic forces. (CO and CN- are strong-field ligands while the Br- and I- are weak-field ligands.) Since there is no single theory for bonding in coordinate compounds that takes into account properties such as color, magnetism, stereochemistry, and bond strength, several different approaches have to be applied. A ligand is a oxygen atom in Fe(H 2 O) 6 3 or a nitrogen atom in Zn(NH3)4 2.ĭ Orbitals: a type of atomic orbital ( regions of space around the nucleus of an atom where an electron is likely to be found) with five distinct shapes they give transition metals the ability to easily give and take electrons.Ĭrystal Field Splitting: The energy difference between two sets of d orbitals in a metal atom when ligands are present.Įx.  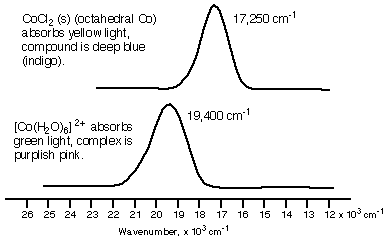
Ligand: A molecule or an ion that is bonded to the metal ion in a complex ion.Įx. The spectrochemical series was first proposed in 1938 based on the results of absorption spectra of cobalt complexes.Ī partial spectrochemical series listing of ligands from small to large is given below.Ĭomplex Ion: An ion containing a central cation bonded to one or more molecules or ions. Rank Dmg In The Spectrochemical Series. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
February 2023
Categories |
 RSS Feed
RSS Feed
